Author: Svetlana Nikic, Founder, Precision Oncology Consulting
With contributions from Carmen Rubio Alarcón (LinkedIn) and Alina Liu (LinkedIn), who supported the evidence review and structuring of the analysis.
Many targeted therapies rely on biomarker identification to select eligible patients, and therefore access to testing and access to treatment are inherently interconnected. As a result, market access strategies should encourage collaborative frameworks between diagnostics developers, pharmaceutical companies, and healthcare systems to ensure that testing translates into actionable treatment decisions. In practice, this includes generating robust real-world evidence on how genomic testing influences clinical decision-making, quantifying therapy uptake following testing, and developing country-specific value dossiers that link testing to downstream clinical and economic outcomes. Aligning the value proposition of testing with the outcomes achieved through matched therapies is critical to strengthening reimbursement discussions and securing sustainable access.
With that in mind, we explore here the key market access challenges associated with broad molecular profiling, specifically Comprehensive Genomic Profiling (CGP), across Europe. Demonstrating clinical utility, often reflected through improved survival outcomes, remains a central requirement for the adoption of innovative molecular diagnostics and continues to be a primary focus for technology developers. However, clinical utility is not determined by the test alone, nor does its demonstration guarantee integration into routine clinical practice.
Beyond evidence generation, successful access and adoption of CGP depend on addressing a broader set of economic, organisational, technical, and societal barriers. These include variability in reimbursement frameworks, infrastructure and workforce readiness, integration into clinical pathways, and alignment with payer expectations. Such challenges are particularly pronounced in Europe’s fragmented healthcare landscape, where heterogeneity across countries continues to shape both the pace and extent of CGP implementation.
The CGP implementation paradox
The CGP-guided targeted treatment administration has demonstrated the potential to improve patient outcomes in several studies across multiple geographies when compared to single gene or small panel testing. A recent study investigating 3884 non-small cell lung cancer (NSCLC) patients confirmed that overall survival (OS) improves when targeted treatment is administered based on actionable alterations patients’ carry. The key point of the study is that the proportion of patients with actionable biomarkers identified increased significantly using CGP testing versus using a small panel (31% vs 14%, p<0.001)1. Similarly, a recent pan-cancer study comparing CGP to an (in silico simulated) 50 gene panel in 3216 patients showed that broad molecular profiling detects significantly more actionable mutations than a smaller gene panel (67% vs 33%, p<0.001)2.
Additionally, CGP enables the detection of emerging, complex, large-scale genomic biomarkers such as tumour mutational burden (TMB) and homologous recombination deficiency (HRD), which cannot be reliably assessed using small gene panels. These biomarkers are particularly relevant in specific clinical contexts, including the use of immunotherapies in tumour types such as NSCLC for TMB, and the identification of patients who may benefit from PARP inhibitors in HRD-associated cancers such as ovarian and breast cancer.
Taken together, the available evidence suggests that CGP has the potential to improve patient outcomes, including survival, particularly when linked to actionable findings and access to appropriate targeted therapies. Yet, reimbursement coverage remains highly fragmented across European healthcare systems. If the test can result in a benefit for patients, why is reimbursement not yet in place? How can this problem be tackled?
The Access Gap: Where does value get lost during reimbursement evaluation?
The impact of CGP on clinical and economic outcomes is not determined solely by test performance or clinical utility. It is also strongly influenced by the availability of targeted therapies, their clinical effectiveness, and country-specific healthcare system factors such as reimbursement frameworks, infrastructure, funding resources, access to molecular tumour boards (TMBs) and / or specialised centres.
Access to therapy:
Even if a diagnostic tool has excellent sensitivity/specificity, identification of genomic alterations may not result in improved survival if the matched therapy is not available. Namely, only 46% of innovative medicines approved centrally in the period 2020-2023 (173 in total) were found to be available to European patients in 2024. Patient access to oncology therapies is influenced by a multitude of factors and remains highly variable across Europe
One influencing factor is the time from EMA approval to reimbursement by national bodies, which ranges from 150 days in Germany, to almost 500 days in Spain, and around 900 days in Poland. Another factor is the speed of adoption of each country. Recent data shows that countries with similar reimbursement timelines can have vastly different patient access levels. For example, despite a short delay from EMA approval to reimbursement, the Netherlands has low access (~22%) during the first year, while France achieves high access (~85%) despite a much longer delay.
While administrative timelines for reimbursement are often cited as the primary metric for patient access, the disparity between the Netherlands and France demonstrates that official approval is only one piece of a much larger puzzle. In France, high initial access is achieved through a robust early-access framework that prioritizes clinical delivery while price negotiations are still ongoing. Conversely, in the Netherlands, uptake is often stifled by “reimbursement locks” and a decentralized budgeting system that places the financial burden on individual hospitals. Beyond these structural hurdles, the “last mile” of access is heavily dependent on the clinical environment: physicians must be sufficiently aware of the new therapy’s benefits, and the diagnostic infrastructure must be in place to identify eligible patients. Without clear national guidelines and the availability of necessary biomarker testing, even a “reimbursed” drug may remain out of reach for the patients who need it most.
Even when innovative therapies are approved and reimbursed, access remains fundamentally constrained if patients are not appropriately identified through biomarker testing. The evidence highlights a critical disconnect in clinical practice: Cancer Patients Europe (CPE) carried out an analysis on patterns of ESR1 and liquid biopsy testing across 1,268 breast-cancer patients in five countries and they found that 70% of patients did not know which biomarkers they have been tested for, and 75% reported no discussion of biomarker testing with their oncologist3. This indicates that the barrier to effective treatment is not only regulatory or financial, but also embedded within the clinical pathway itself. Without systematic integration of comprehensive genomic profiling and clear physician–patient communication, approved targeted therapies cannot reach the patients most likely to benefit, limiting their real-world impact and reinforcing inequities in access.
These factors, together with other context-specific circumstances, translate in an “access gap” between the number of patients with actionable mutations identified by CGP, and the number of patients that can access the matched targeted therapy, which ultimately leads to a loss in the value of the test.
Table 1 shows an overview of recent pan-cancer studies showing this access gap across several regions.
Table 1: The access gap.
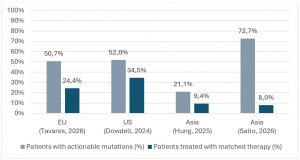
Table 1: Comparison of the percentage of patients with actionable mutations identified using CGP and the percentage of patients receiving the matched therapy. [2,4-6]
The consequence of this access gap for reimbursement of CGP testing is that the clinical utility of the test is underestimated when evaluating real world evidence (RWE). This becomes evident in studies directly comparing the survival between patients harboring actionable mutations and receiving the matched targeted therapy versus those with actionable mutations that do not receive the treatment. A recent pan-cancer study investigating 1482 patients with advanced cancer undergoing CGP testing in the US showed a benefit in OS for patients treated with CGP-guided therapy versus patients treated with chemotherapy (median OS= 25 vs 17 months, HR= 0.66 [0.52-0.84], p<0.001)4. An European pan-cancer study in patients with advanced cancer using disease control at 16 weeks as surrogate endpoint for clinical utility showed that 60% of the patients receiving CGP-guided therapy remained progression free at the selected timepoint5. A study from Asia with 456 patients with advanced or locally advanced diasease tested with CGP showed, once again, that OS was significantly improved in patients with actionable mutations receiving CGP-guided matched targeted therapy compared to patients not receiving the matched therapy (median OS= 26 vs 11 months, HR=0.28 (0.14-0.55), p<0.001)6. These findings demonstrate that across diverse geographies and study designs, the evidence consistently shows that CGP-guided therapy is associated with clinically meaningful improvements in patient outcomes, including significantly longer OS and sustained disease control compared with non-matched or standard treatment approaches.
Response to therapy:
Not all patients respond to a matched targeted treatment, and that affects the impact in survival of the test.
As discussed above, the clinical impact of a molecular diagnostic test cannot be assessed in isolation. The value of identifying an actionable genomic alteration is intrinsically linked not only to the availability, but also to the effectiveness of the corresponding targeted therapy. It is important to highlight that the presence of a biomarker does not guarantee a uniform therapeutic response. Even among patients with the same genomic alteration and within the same tumour type, responses to targeted therapies can vary considerably due to tumour heterogeneity, co-occurring genomic alterations, and differences in disease biology7. Consequently, the real-world clinical benefit of molecular testing is ultimately determined by the complex interaction between biomarker detection, treatment efficacy, and patient selection. This adds an extra layer of complexity when evaluating reimbursement and highlights the need for value-based reimbursement evaluations alongside traditional cost-based ones.
Context differences among different healthcare systems:
Health technology assessment (HTA) frameworks for molecular diagnostic tests remain highly heterogeneous across Europe. Evaluation criteria, methodological approaches, and the relative weight assigned to different evidence domains vary substantially between countries. While some systems place stronger emphasis on analytical and clinical validity, others prioritise demonstrated clinical utility, budget impact, or broader system-level considerations such as care pathway optimisation. In 2025, we have created and published insights8, focused on navigating this complexity, providing a structured, cross-country perspective on evidence expectations and stakeholder engagement into clear, actionable strategies for market access and adoption.
In addition, economic evaluations of genomic testing are particularly sensitive to contextual factors that differ between healthcare systems. Variables such as the price of matched targeted therapies, reimbursement structures, access to MTBs, and the proportion of patients who ultimately receive biomarker-guided treatment can significantly influence the perceived value of testing. Consequently, the cost-effectiveness of CGP may differ considerably across countries depending on treatment costs and the real-world uptake of matched therapies. For example, a cost-effectiveness study evaluating the use of CGP versus a small gene panel showed that the estimated incremental cost-effectiveness ratio (ICER) of CGP versus SP was $174,782 and $63,158 per life-year gained in the US and Germany, respectively, suggesting that the cost-effectiveness of CGP is highly context-dependent9.
The absence of a harmonised assessment framework means that innovative diagnostic technologies must typically undergo separate evaluations in each national system. In decentralised healthcare environments such as Italy and Spain, additional regional review processes may apply. As a result, the pathway from regulatory approval to routine clinical adoption often becomes fragmented and prolonged, delaying patient access to precision oncology innovations.
How to overcome these barriers: Integrated market access plan
An effective market access strategy for molecular diagnostics should address testing implementation and therapy access in parallel (as done in Japan). In precision oncology, the value of a biomarker test is realised only when patients identified through testing can access the appropriate targeted treatment. Therefore, market access planning must anticipate both operational barriers to testing and reimbursement challenges related to downstream therapy utilisation.
A pragmatic access strategy can be structured around three complementary pillars.
Establish a robust testing implementation strategy
- The initial focus should be on priority tumour types where clinical evidence and guideline support are already well established, such as NSCLC. Clear integration of the test within existing diagnostic pathways is essential, including alignment with current pathology workflows, sample requirements, and turnaround time expectations.
- In parallel, laboratories must be supported in implementing and validating the technology in accordance with recognised quality standards (for example, ISO 15189 and EQAs defined by IQN Path), while ensuring operational feasibility and sustainable testing capacity. Demonstrating that the test can be implemented reliably within real-world clinical workflows is often a prerequisite for payer confidence.
Strengthen the role of Molecular Tumour Boards
- MTBs play a central role in translating complex genomic results into clinically actionable treatment decisions. However, some studies show how in some countries MTB are not yet consistently established5. Emphasizing the importance of MTBs, and supporting them through standardised reporting frameworks, structured interpretation guidelines, and decision-support tools can improve consistency in how genomic findings are applied in clinical practise.
- Equally important is the establishment of metrics to capture MTB-driven treatment decisions and associated outcomes derived from the use of CGP (e.g. number of targeted therapies recommended using CGP vs SoC, cost savings derived from high cost SoC therapies not given). These data can provide valuable real-world evidence to inform reimbursement discussions and demonstrate the clinical impact of biomarker-driven care. Continuous education and training for oncologists, pathologists, and other healthcare professionals are critical to ensure that genomic information is interpreted appropriately and used consistently across centres.
Concluding remarks
Demonstrating strong analytical performance and clinical utility alone is rarely sufficient to secure the routine adoption of a molecular diagnostic test. In the European context, successful integration into clinical practise depends equally on addressing system-level considerations such as reimbursement pathways, laboratory implementation, clinical workflow integration, and access to matched therapies.
To achieve sustainable reimbursement and facilitate timely implementation across European healthcare systems, developers should adopt an integrated market access strategy that is tailored to the specific requirements of each country. This includes early engagement with key decision-makers, proactive evidence planning aligned with payer expectations, and a clear implementation roadmap that addresses both clinical and operational barriers to adoption.
References:
- Jeanna Wallenta Law et al.Real-World Impact of Comprehensive Genomic Profiling on Biomarker Detection, Receipt of Therapy, and Clinical Outcomes in Advanced Non–Small Cell Lung Cancer. JCO Precis Oncol8, e2400075(2024). DOI:10.1200/PO.24.00075
- Dowdell AK, Meng RC, Vita A, et al. Widespread Adoption of Precision Anticancer Therapies After Implementation of Pathologist-Directed Comprehensive Genomic Profiling Across a Large US Health System. JCO Oncol Pract. 2024;20(11):1523-1532. doi:10.1200/OP.24.00226
- Advancing precision oncology treatment and testing across Europe: An evidence-based roadmap for healthcare system stakeholders to improve cancer care. Efpia, Mar 2026.
- Saito Y, Horie S, Kogure Y, et al. Real-world clinical utility of comprehensive genomic profiling in advanced solid tumors. Nat Med. 2026;32(2):690-701. doi:10.1038/s41591-025-04086-8
- Tavares N, Simões P, Lopes-Brás R, Pacheco TR, Damaso S, Mansinho A, Ribeiro LA, Nogueira-Costa G, Abreu C, Barroso T, et al. Assessing the Impact of Comprehensive Genomic Profiling on Therapeutic Selection for Advanced Solid Tumors in Portugal. Current Oncology. 2026; 33(2):66. https://doi.org/10.3390/curroncol33020066
- Hung LJ, Huang CY, Tung KC, et al. Comprehensive genomic profiling in multiple cancer types: A comparative analysis of the National Biobank Consortium of Taiwan and clinical practice cohorts. J Formos Med Assoc. 2025;124(9):830-838. doi:10.1016/j.jfma.2024.09.001
- Zhu L, Jiang M, Wang H, et al. A narrative review of tumor heterogeneity and challenges to tumor drug therapy. Ann Transl Med. 2021;9(16):1351. doi:10.21037/atm-21-1948
- https://precision-oncology-consulting.com/precision-oncology-insights/
- Spencer S, Ye W, Peng S, Zou D. Cost-Effectiveness Analysis of Comprehensive Genomic Profiling in Patients with Advanced Non-Small-Cell Lung Cancer Using Real-World Data. J Mol Diagn. 2025;27(9):850-858. doi:10.1016/j.jmoldx.2025.05.011
